Introduction
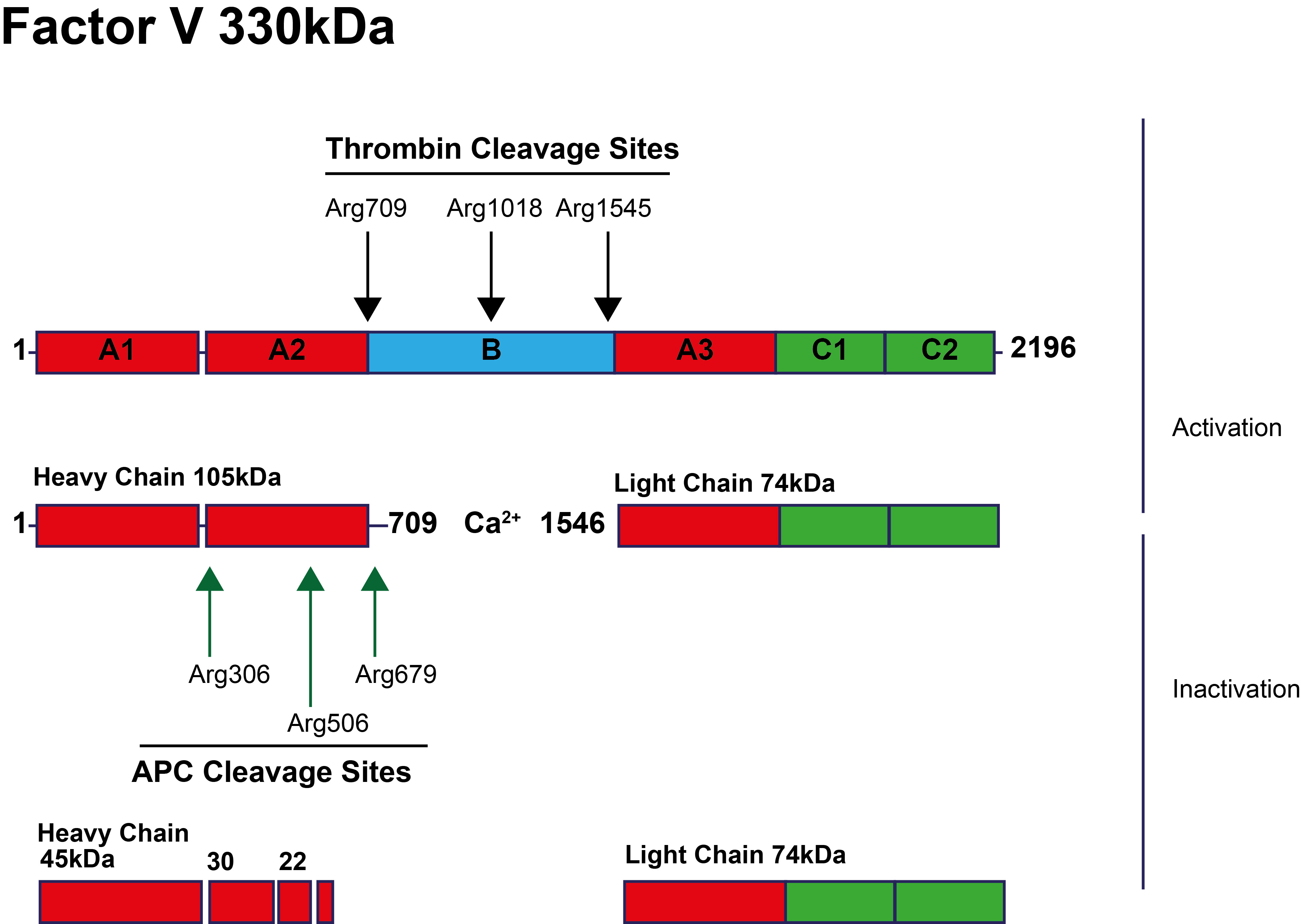
Resistance to Activated Protein C [APC-R] was first reported in 1995 and in approximately 90% of cases is due to the Factor V Leiden [FVL] mutation – a G1691A missense mutation within the F5 gene resulting in the replacement of the Arginine 506 residue by a Glutamine [R506Q] and the abolition of an Activated Protein C [APC] inactivation cleavage site in Factor Va. This amino acid substitution slows the rate of FVa inactivation by approximately 10-fold, resulting in increased Thrombin generation and a hypercoagulable state.
The first cleavage of FVa at Arg506 results in partial inactivation of FVa but is essential for exposure of the other APC cleavage sites in FVa at Arg306, Arg679 and Lys994 and the inactivation of FVa. The inactivation of FVa also leads to reduced cofactor activity for the APC-mediated inactivation of Factor VIIIa.
This is illustrated diagrammatically below:
[APC - Activated Protein C; A1-A3, B & C1-C2 represent the domains of Factor V].
Principles & Methodology
A number of tests have been developed to screen for APC resistance [APC-R]:
| Method | Explanation |
|---|---|
| APTT | The original test for APC resistance [APC-R] involved measuring the APTT of a plasma sample with or without the addition of exogenous APC. In a plasma sample without APC-R, the addition of APC inactivates Factor Va and Factor VIIIa and so prolongs the clotting time of the APTT. In contrast in a sample with the Factor V Leiden mutation, the prolongation in the clotting time is less. A ratio is derived from: [APTT+APC]/[APTT-APC] A limitation of this test is that it requires a normal APTT in the patient and so cannot be used in cases in which there is a prolongation of the APTT e.g. patients on oral anticoagulants or with a Lupus Anticoagulant. In addition deficiencies of Factor VIII, IX, XI or XII that prolong the APTT may generate false positive APC-R results. Polybrene may be included in some assays. Polybrene can neutralise Heparin [<1 IU/mL] and therefore removes this anticoagulant from the assay. Individuals without the FVL mutation generally have a ratio of >2.0 and individuals who are heterozygous for the FVL mutation have a ratio <2. However, there is considerable overlap between healthy individuals and heterozygotes and individual laboratories mush establish their own cutoff values. This test is also useful for detecting acquired APC-R and which represents an independent risk factor for venous thrombosis. In addition the test will detect other causes of APC-R arising from mutations within the F5 gene other than the FV Leiden mutation. |
| Normalised APTT | The normalised APTT was established to try and improve the discrimination between FVL heterozygotes and individuals without the FVL mutation i.e. a normal Factor V. The normalised ratio is derived by first dividing the APTT in the presence of APC by the APTT in the absence of APC [as with the original APTT screening test above.] This ratio is then normalized against a reference pool to obtain an APC sensitivity ratio. [APTT+APC]/[APTT-APC] of patient sample [APTT+APC]/[APTT-APC] of a normal reference plasma pool 80% of patients with an APC sensitivity ratio <0.84 and 100% of patients with an APC sensitivity ratio <0.70 were heterozygous or homozygous respectively for the FVL mutation. |
| Modified APTT with Pre-dilution in Factor V-deficient plasma | This is a modification of the original APTT screening test in which a
pre-dilution [1 +4] of patient plasma with Factor V-deficient plasma is made before the addition of APC and calcium. The normal range for an APTT-based assay for APC-R is higher in the newborn infant compared with older children and adults. It decreases to the adult level by 6 months of age. A modification of the assay for the use in infants <6 months of age involves a 1:11 dilution of patient plasma in Factor V deficient plasma, in contrast to the 1:5 dilution for adults. The modified APTT assay reduces the number of exogenous confounding factors that might affect the APTT e.g. High FVIII levels, Vitamin K antagonists, Pregnancy, the Oral Contraceptive Pill - and makes the test specific for mutations within FV. The presence of a Lupus Anticoagulant, by competing for Phospholipid, can prolong the APTT measurements in these assays and are a major source of false-positive results if the test is used as a screening test for the FVL mutation. Protein C antibodies may also interfere with this test. It is important to remember that this modified assay is specific only for mutations within Factor V whereas the original APTT assay without Factor V–deficient plasma pre-dilution, measures APC-R from any cause. In some cases this assay also contain polybrene which makes it insensitive to Unfractionated Heparin [UFH] and Low Molecular Weight Heparin [LMWH] although high heparin levels may overcome the neutralising effect of the polybrene leading to a falsely elevated APC-R ratio. The test is sensitive to the presence of the direct Thrombin Inhibitors and some direct Factor Xa Inhibitors. |
| Pefakit® APC-R Factor V Leiden screen | The Pefakit® APC-R Factor V Leiden screen is a plasma-based functional
clotting assay and differs from other functional APC
resistance tests by using a specific FV activator isolated from
a snake venom (RVV-V from Daboia russelli). The test is performed with and without the addition of APC and involves . Coagulation is triggered by the addition of a Factor V dependent Prothrombin activator - [Noscarin isolated from the venom of a snake Notechis scutatus scutatus] - in the absence of calcium. The time for clot formation is recorded and the ratio: (Clotting Time + APC/Clotting Time - APC] determined. If the FVa molecules have been eliminated during the incubation step, the velocity of Prothrombin activation by the FV-dependent Noscarin is slow and therefore, the clotting time is prolonged. The reagents employed in this test contain polybrene which makes it insensitive to Heparin up to a concentration of <2 IU/ml (UFH and LMWH) or Pentasaccharide levels <2 µg/ml. The test is insensitive to the presence of |
| Chromogenic Factor X Assay | The chromogenic assay for detecting APC resistance is based on the capacity of APC to limit the generation of Factor Xa by inactivating Factor Vllla in plasma. The ratio of the Factor Xa amidolytic activity in a sample without APC to its Factor Xa activity with the addition of APC reflects the response of the plasma coagulation system to APC. |
| Russell Viper Venom Assay | This test is based on the dilute Russell Viper Venom Time (dRVVT). The dRVVT is prolonged when a plasma sample is pre-incubated with a diluted snake venom isolated from Agkistrodon contortrix contortrix which activates Protein C. The test result is expressed as the ratio between the dRVVT with and without addition of the venom. The reagents contain a high phospholipid concentration which reduces their sensitivity to Lupus Anticoagulants and in addition a heparin neutraliser. Pre-dilution of the test plasma sample in Factor V deficient plasma allows the distinction between controls and patients with deficiencies of Protein C, Protein S, Antithrombin and the Factor V Leiden mutation. |
| Thrombin Generation-Based Assays | The Endogenous Thrombin Potential [ETP]-based
APC-R assay is based on the measurement of Thrombin
generation in the presence and in the absence of a defined amount
of exogenous APC. In a normal plasma sample that contains no exogenous APC, the Thrombin generation curve is dependent upon the variables [both pro- and anticoagulant] that regulate Thrombin formation. However, in the presence of APC, in a normal plasma sample, Thrombin generation is significantly decreased. The amount of APC added to the assay is based upon a 90% decrease in the ETP between the two assays i.e. the ETP results with and without the addition of APC. Results are expressed as a ratio - the normalised APC sensitivity ratio (nAPCsr), which is derived from the ratio of the ETP measured in presence and absence of APC in the plasma sample under investigation divided by the same ratio of the reference plasma.  |
Interpretation
The interpretation of the test is very much dependent upon the specific test. Variables that have been shown to affect the original APTT-based test include:
Reference Ranges
These very much depend upon the specific test. A test ideally should be able to differentiate normal individuals without APC-R from individuals who are heterozygous or homozygous for the FVL mutation.
What Test Next
Individuals with a low APC ratio usually go on to have a screen of their F5 gene for the Factor V Leiden mutation. APC resistance independent of the FV Leiden mutation appear to a risk Factor for VTE. Some F5 mutations associated with APC-R but which are not due to the FVL Arg506Gln mutation have also been reported:
| Factor V Variant | Nucleotide substitution | Mutation | Location |
| Factor V Leiden | G1691A | Arg506Gln | Exon 10 |
| Factor V Cambridge | G1691C | Arg306Thr | Exon 7 |
| Factor V Hong Kong | A1090G | Arg306Gly | Exon 7 |
| Factor V NARA | W1920R | Trp1920Arg | Exon 20 |
| Factor V Liverpool | T1250C | Ile359Thr | Exon 8 |
| Factor V Bonn | C4070G | Ala540Val | Exon 10 |
Click HERE to return to the top of the page.
