Introduction
The Von Willebrand Ristocetin Cofactor [vWF:RCo] assay measures the ability of a plasma sample to agglutinate platelets in the presence of the antibiotic Ristocetin. The rate of Ristocetin induced agglutination is related to the concentration and functional activity of the plasma von Willebrand factor.
Two binding sites for Ristocetin have been identified within VWF and which flank the platelet-binding A1 domain: Cys1237–Pro1251 and Glu1463–Asp1472. Both sites are proline rich and brought into proximity by a disulfide bond between Cys1272 and Cys1458.
Ristocetin Induced platelet agglutination (RIPA) is similar to the Ristocetin Cofactor Assay but RIPA measures platelet agglutination induced by Ristocetin-mediated VWF binding to the platelet Gp1b receptor but in this case the Ristocetin is added directly to the patient platelet rich plasma and there are no serial dilutions of the plasma sample. Low dose RIPA is used to screen for Type 2B VWD [and platelet Type VWD] as platelets from such individuals will agglutinate at low concentrations of Ristocetin but normal platelets will not.
The ISTH recommendations for von Willebrand Factor Activity assays are summarised below:
| Abbreviation | Description |
|---|---|
| VWF:RCo | An assay that measures VWF activity and employs platelets and Ristocetin in the assay |
| VWF:GPIbR | An assay that is based upon the Ristocetin-induced binding of VWF to a recombinant wild-type GPIb fragment |
| VWF:GPIbM | An assay that is based upon the spontaneous binding of a gain-of-function mutant GPIbα fragment |
| VWF:Ab | An assay that is based upon the binding of a monoclonal antibody to a VWF A1 domain epitope. |
A number of methods exist for measuring VWF:RCo and these are summarised below:
Principles & Methodology
a. Platelet Agglutination method
The method is similar to a factor assay and was for many years the only method available for measuring VWF activity [VWF:RCo]. The test has a relatively poor sensitivity which makes it unsuitable for measuring VWF levels <10 IU/dL. In addition the high CV of the assay due to a number of variables including the source of Ristocetin and the platelets used in the assay, may lead to problems with the diagnosis of VWD. Furthermore, two sequence variations in the VWF gene [p.P1467S and p.D1472H] that encode the A1 domain can lead to defective binding of Ristocetin to VWF leading to a low VWF:RCo although the sequence variations are not associated with a bleeding disorder.
There are rare cases of patients with mutations in the A3 domain of VWF that are associated with impaired binding to collagen. The VWF:CB/Ag ratio is abnormal but the normal VWF:RCo assay is normal.
| Step | |
|---|---|
| 1 | Fresh, formalin-fixed or commercial lyophilised fixed platelets are mixed with dilutions of patient plasma, a fixed concentration of Ristocetin and light transmission aggregometry is performed. |
| 2 | Patient plasma samples are normally performed in duplicate at a single dilution but a second dilution may be required depending upon the VWF concentration in the plasma sample. |
| 3 | Agglutination is allowed to proceed and from the traces the slope of the curve is derived. A standard curve is established by using a similar method but replacing the patient plasma with serial dilutions of normal plasma. |
| 4 | The slope of the curve is plotted against dilution on double-log paper and a straight line obtained. From this the Ristocetin cofactor activity in the patient’s plasma samples can be derived. The following traces demonstrate this more clearly: 
Click HERE to see a larger view of this image/illustration. The slope is calculated by plotting the tangent of the slope and them measuring the distance from the origin to where the tangent crosses the X or Y axis. The distance on the Y axis is then divided by the distance on the X-axis and the slope derived. For example if for the normal plasma sample, the distance from the origin to where the tangent intercepts the X-axis is 12mm and the distance on the Y axis is 40mm then the slope is 40/12 = 3.3. |
| 5 | The slopes (S) of each of these traces derived from serial dilutions of normal plasma are plotted against the dilution on double-log paper. A straight line is drawn through the points and by comparison again the reference plasma the VWF:RCo activity of an unknown plasma sample can be calculated - see below: Click HERE to see a larger view of this image. In Plasma Sample - 1, there is no aggregation [more correctly no agglutination] and so the VWF:RCo activity is <1 IU/dL. In Plasma Sample - 2, the slope is 0.75 and so from the standard curve we can calculate that this equates to a VWF:RCo activity of ~11 IU/dL - the Reference plasma has a VWF:RCo concentration before dilution of 100 IU/dL. We can check that the VWF:RCo concentration in Plasma Sample 2 is correct because the slope of Plasma Sample 2 lies between that of the 1/8 dilution [vWF concentration 12.5 IU/dL] and the 1/16 dilution [vWF concentration 6.25 IU/dL] of the reference plasma. Finally - we are using undiluted plasma in Plasma Samples 1 and 2 and so there is no requirement to make any corrections for dilution. |
It is possible to replace the aggregometer with a microtitre plate or an ELISA plate reader but the principles remain the same.
The platelet-based method for measuring VWF:RCo is time-consuming and is no longer widely performed.
b. ELISA assays that assess direct binding of plasma VWF to platelet GPIb
In these assays the GPIb can be of one of two sources:
a. A
recombinant GPIb fragment. Briefly, an ELISA plate is coated with an anti-GPIb monoclonal antibody after which the recombinant GPIb fragment is added. VWF is then added [either the reference plasma or the test sample] to the GPIb-coated wells in the presence of a fixed concentration of Ristocetin. VWF in the plasma sample binds to the immobilised GPIb fragment and the bound vWF is detected with an anti-human VWF horseradish peroxidase [HRP] labelled antibody. The bound antibody is quantified in a colorimetric reaction by adding the HRP substrate OPD [1,2 ortho-phenylendiamine dihydrochloride] and measuring the absorbance at 492 nm. The absorbance is proportional to the concentration of VWF. A calibration curve is constructed by using serial dilutions of a reference plasma against which the absorbance of the test plasma is measured and from which the VWF:RCo activity can be derived.
b. A proteolytic fragment termed 'glycocalicin' consisting of the extracellular part of the α-chain of the platelet GPIb receptor, which can be removed from the platelet surface by treatment with various proteases. interestingly glycocalicin is also found in normal plasma and can be purified to be used in the assay and was the source of the glycocalicin in the original assay.
In the glycocalicin VWF:RCo assay, microtitre plates are incubated with an anti-GPIbα monoclonal antibody and after washing the plates are saturated with glycocalicin. Plasma [controls, tests and standard] and a fixed concentration of Ristocetin are added and the bound vWF is detected with an anti-human VWF horseradish peroxidase [HRP] labelled antibody. The bound antibody is quantified in a colorimetric reaction by adding the HRP substrate OPD [1,2 ortho-phenylendiamine dihydrochloride] and measuring the absorbance at 492 nm.
c. Flow Cytometry
VWF:RCo can also be measured by flow cytometry. In this method a mixture of green and red fluorochrome-labeled and formalin-fixed platelets are incubated with plasma samples in the presence of Ristocetin. Each VWF multimer has multiple binding sites for the platelet
GPIb receptor. When both green and red
fluorescently-labeled platelets bind to a single VWF molecule, a microaggregate forms. The extent of platelet microaggregates, reflected by the double positive (green
and red) events, correlates with plasma VWF:RCo activity. The extent of the microaggregate
formation is affected by the binding affinity of VWF to platelets
and the molecular weight of the VWF multimer. There are no double positive events if the VWF
multimer molecular weight is too low to bind at least two
platelets.
d. Latex-based Assays
This assay used a recombinant GPIb alpha fragment [rGPIb] which contains the VWF-binding site and which is coated onto latex particles using a highly specific monoclonal antibody. If VWF is present in the sample then it binds to the GPIb alpha fragment and in the presence of Ristocetin causes agglutination of the latex particles. The degree of agglutination is directly proportional to the activity of VWF in the sample and is determined by measuring the decrease in light transmission caused by the aggregates.
e. Chemiluminescence Assays
This assay employs a recombinant GPIb alpha fragment [rGPIb] that is coated onto magnetic particles through a specific monoclonal antibody which orientates the rGPIb fragment to facilitate binding to VWF after incubation with a Ristocetin-containing buffer. The bound VWF is then detected by chemiluminescence using an isoluminol-labelled anti-VWF antibody. The degree of light emitted is proportional to the VWF:RCo activity.
Interpretation
The measurement of VWF activity is fundamental to the diagnosis of VWD but the VWF:RCo assay has a number of potential problems:
i. A
high intra-laboratory and inter-laboratory variation with the potential for falsely elevated high or low values.
ii. A lower limit of detection in the region of 10-20 IU/dL.
iii.
There are in addition VWF variants [e.g. Asp1472His] that affect the ability of Ristocetin to bind to VWF leading to an artefactually low VWF:RCo assay but which do not affect VWF function.
|iv.
It does not actually measure the physiological function of VWF.
In the most recent assays, platelets have been replaced by rGPIb fragments immobilised onto either an ELISA plate or to latex particles allowing the accurate measurement, in general, of VWF activity in the range of 1-10 IU/dL - see Epitope-specific Assays.
In individuals with normal VWF structure and function, the results of VWF:RCo and VWF:Ag assays are similar.
Reference Ranges
The reference range for VWF:RCo is usually in the region of 50-150 IU/dL. Von Willebrand factor is an acute phase protein and so the levels may rise at times of stress e.g. in pregnancy.
What Test Next
Measurement of VWF activity is usually undertaken as part of a screen to establish or exclude a diagnosis of von Willebrand disease. These assays are also used to monitor replacement therapy in individuals with VWD.
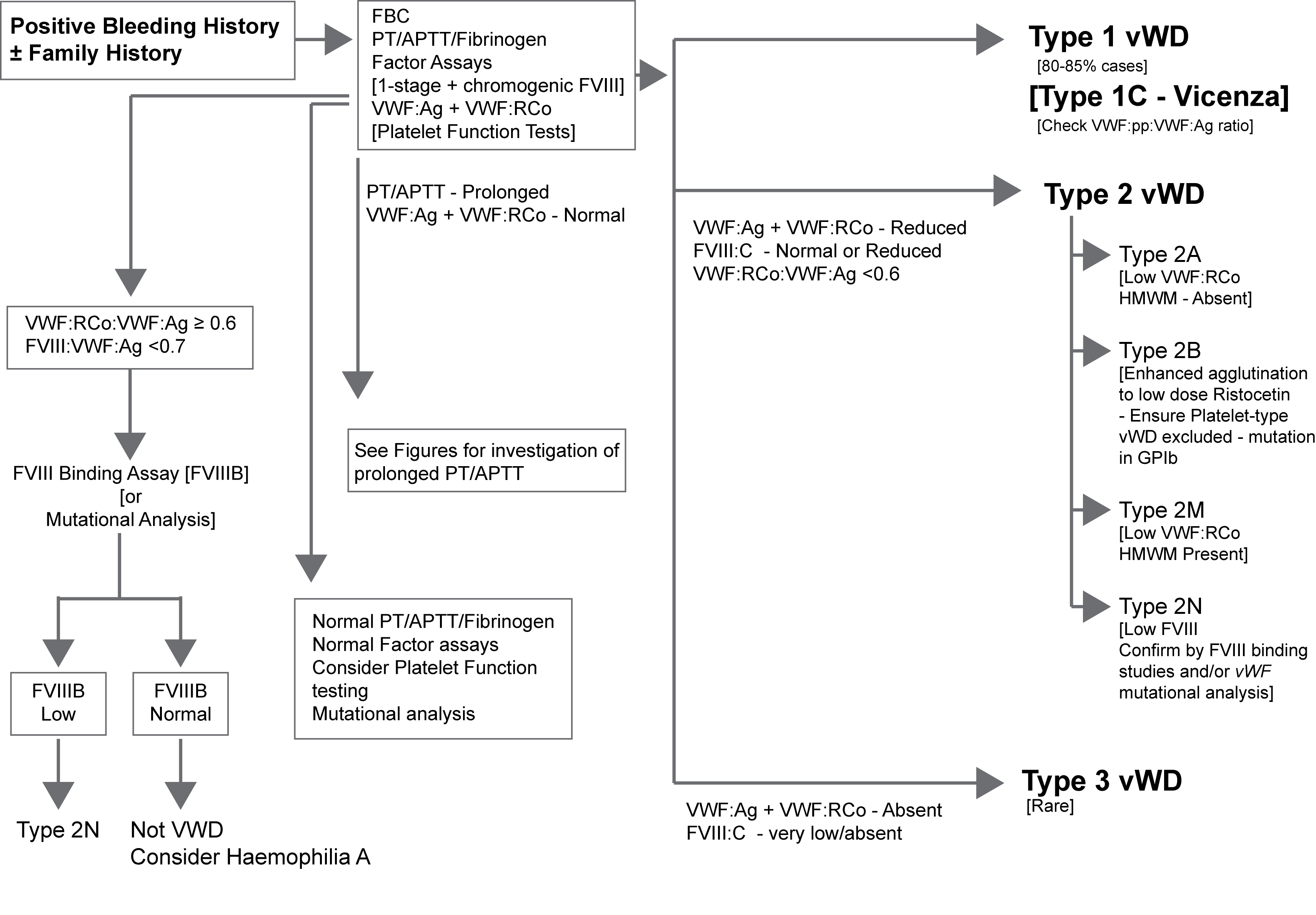
The illustration below outlines one approach to the diagnosis of suspected VWD. Click HERE for an expanded version of this illustration.