Introduction
The generation of Thrombin [Factor IIa] is a fundamental part of the clotting cascade and as such an estimation of an how well a particular individual can generate Thrombin may correlate with either a risk of bleeding or thrombosis. Historically we have analysed coagulation using tests such as the PT and APTT and although widely performed they have a number of intrinsic problems:
- they look at the clotting cascade in isolation
- they are non-physiological
- they show a poor correlation with the clinical phenotype i.e. the PT or APTT may be prolonged but this does not necessarily predict the bleeding phenotype
- they are, in general, insensitive to prothrombotic states
Furthermore the PT and APTT use the formation of a fibrin clot as the endpoint of the test and yet we know that this occurs when only ~5% of the total amount of Thrombin that is generated, occurs.
Principles & Methodology
1. Thromboplastin Generation Tests [TGTs]: The original Thrombin generation assays published in 1953 were time consuming and technically difficult to perform requiring sub-sampling of reaction mixtures at regular intervals into secondary tubes containing Fibrinogen and which allowed clotting to proceed. The clotting times of the Fibrinogen solutions were used to estimate Thrombin concentration after calibration against a Thrombin standard. These so-called 'Thromboplastin generation tests [TGTs'] formed the basis of the 2-stage Factor assay.
This method was simplified some years later but remains in essence a 2-stage assay.
2.
Chromogenic Assays: The Endogenous Thrombin Potential [EPT] was a term introduced by Hemker in 1986 and refers to the total amount of Thrombin generated during the test.
In this test the generation of thrombin is monitored using a chromogenic substrate.
Ordinarily Thrombin is inactivated by Antithrombin and α2-macroglobulin and whilst Thrombin bound to Antithrombin is inactive [it binds to the active site of Thrombin] Thrombin that binds to α2-macroglobulin retains activity to the chromogenic substrate as its binding is to the exosite of Thrombin and so it can continue to cleave the chromogenic substrate. For these reasons a complex algorithm was employed to calculate free Thrombin generation and to remove the contribution from Thrombin bound to α2-macroglobulin.
This test required defibrination of the plasma and it is now recognised that this can have a significant effect upon the results obtained from Thrombin generation assays.
3. Fluorogenic Assays: The replacement of the chromogenic substrate with a slow acting fluorogenic substrate enabled the continuous measurement of Thrombin generation and required no prior defibrination of plasma. Further more the signal from the fluorogenic substrate was not quenched by turbidity and so Thrombin generation assays could now be performed in platelet rich plasma.
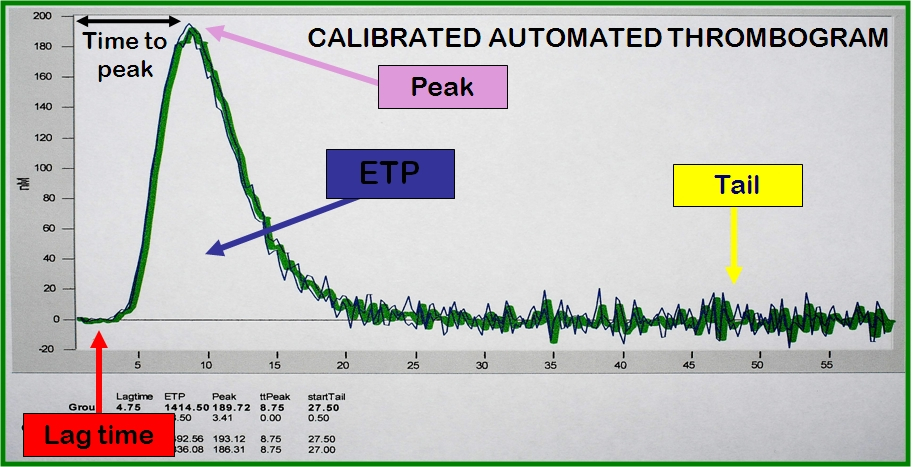
However, the use of a fluorogenic substrate resulted in additional problems as there is no direct correlation between Thrombin activity and fluorescent signal intensity. To over come this, the splitting of the fluorogenic substrate is compared to a constant known Thrombin activity in a parallel non-clotting sample the so called Calibrated Automated Thrombogram [CAT].
Variables
Commonly measured variables when analysing thrombin generation include:
The Lag Time
The Time to Peak Thrombin Generation
The Peak
The Endogenous Thrombin Potential [ETP] - the area under the curve - the total amount of Thrombin generated.
The Tail
The initiation of Thrombin generation corresponds to the clotting times seen with the PT and APTT. In contrast the amplification/propagation stages and the subsequent inhibition of Thrombin correlate with the inhibitors of Thrombin generation both directly and indirectly such as Protein C, Antithrombin and
α2-macroglobulin.
Reference Ranges
Reference ranges vary depending upon the precise methodology employed and so should, therefore, be derived for individual laboratories.
What Test Next
Thrombin Generation Assays are moving in some labs into routine practice and are used in an increasing number of areas.
